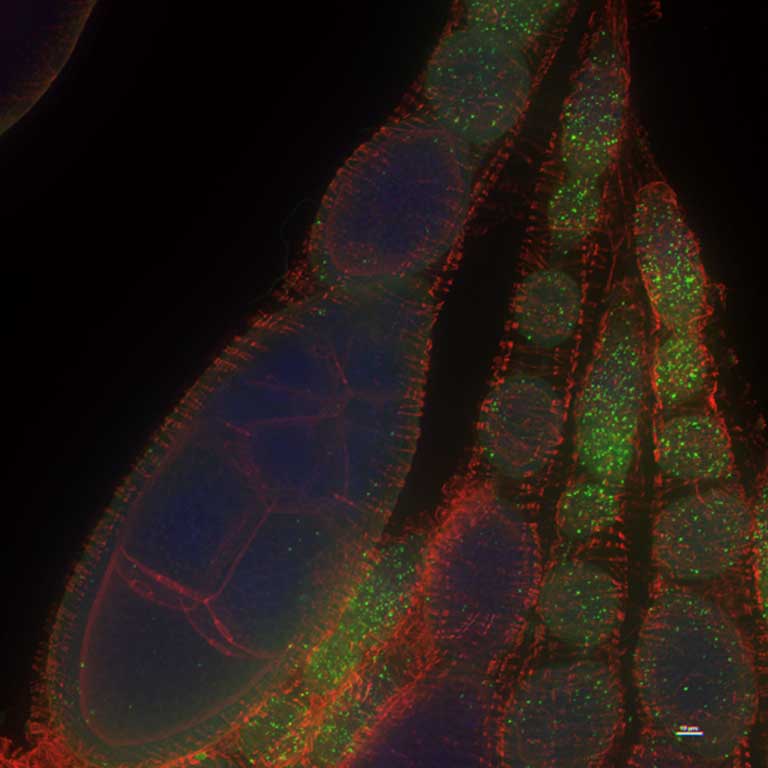
Wolbachia are known as intracellular endosymbionts, meaning that they can only survive within the cells of a host organism. Mosquitoes infected with Wolbachia (either naturally or artificially) have dozens or even hundreds of the bacteria in their cells, tinkering with their biology from within.
But how exactly does Wolbachia alter the cell and infect its host? That’s the question that led Professor Irene Newton to create her lab at IU in 2011.
In her February 2023 paper, currently in the process of peer review, Prof. Newton explored the function of a protein secreted by Wolbachia called WalE1. Newton’s previous work had shown that WalE1 wasn’t a typical bacterial protein. It has a sequence of amino acids called a synuclein domain: something that only exists in vertebrates.
Wolbachia itself clearly isn’t a vertebrate and neither are the insects that it infects, yet it still makes a protein with a synuclein domain. As Newton describes it, this seemingly useless sequence is a genius way of interacting with the host cell.
“Bacterial proteins have certain structures and homologies that look like those from eukaryotic cells,” said Newton, “it‘s like having the right wrench to interact with the right bolt.”
WalE1’s synuclein domain allows it to interact with the host cell precisely and without being detected as an invader. It is the wrench that fits perfectly onto the host cell’s bolt. Only one problem: what is its bolt?
To figure out what exactly WalE1 was targeting in the host cell, Newton and research associate MaryAnn Martin modeled a Wolbachia infection in fruit flies.
Fortunately for them, the IU Bloomington Drosophila Stock Center (the world’s largest Drosophila collection, providing genetic strains of fruit flies across the world) is just steps away. “For our research it’s especially great,” said Newton. “Because of the great tool sets and all the resources here at IU, we can use those resources to translate what we find in flies to the mosquito vector.”